Original: $750.00
-65%$750.00
$262.50The Story
INTENDED USE
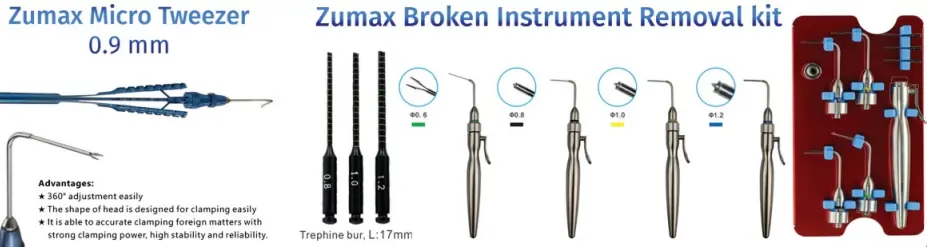
BTR PEN, together with a compatible nitinol loop tip, is designed to remove fragments of broken endodontic instruments from the root canal.
HANDLING DENTAL TOOLS MADE OF STAINLESS STEEL
The instrument is made of stainless steel. Special processes have been undertaken during the manufacture of the dental instruments to ensure corrosion-resistance. In spite of this, the durability of these dental instruments
depends on their proper use and correct maintenance.
HANDLING OF THE BTR PEN
The stainless steel instrument is designed for multiple use, while the BTR PEN-compatible nitinol loop tip is intended for single use. Before each use, the instrument should be inspected for contamination, damage to the material structure (cracks, bends, splits, scratches – deeper scratches). Failure to adhere to the above stated instructions decreases instrument durability and reduces instrument lifetime.
HANDLING WITH BTR PEN NITINOL LOOP TIP
The BTR TIP is delivered non – sterile. Before use, the tip should be sterilized, using standard bags according to the sterilizaction in autoclave. Store in a dry and clean place for storage. Observe the instructions of the autoclave manufacturer. BTR TIP is for single use.
STERILIZE WITHOUT THE PROTECTIVE CUP!
Used tips disposed of responsibly or returned to the manufacturer.
HANDLING THE BTR PEN CALIBRATOR
The calibrator is designed to be reusable. It should be disinfected with isopropyl alcohol and inspected before each use. A blunted or bent calibrator needle can make it difficult to accurately calibrate the nitinol loop, which negatively affects the instrument performance.
CLEANING, RINSING AND WASHING OF BTR PEN:
The packaged instruments are non – sterile. Instruments that are brand new should be thoroughly cleaned with a non-scratching soft bristle brush and an instrument cleaning solution (detergent: ANIOSYME X3; solution prepared according to the manufacturer’s instructions). Ultrasonic cleaning is also recommended.
After cleaning, all components should be rinsed with purified water and dried, as moisture can adversely affect the effects of sterilisation. Instruments must be cleaned, rinsed and dried again and sterilised before each use on a subsequent patient.
STERILIZATION
All (reusable) dental instruments must be disinfected, then washed out and then dried before sterilisation. BTR PEN must always be disassembled before sterilisation. Instruments should be sterilized in accordance with national requirements.
Description
INTENDED USE
BTR PEN, together with a compatible nitinol loop tip, is designed to remove fragments of broken endodontic instruments from the root canal.
HANDLING DENTAL TOOLS MADE OF STAINLESS STEEL
The instrument is made of stainless steel. Special processes have been undertaken during the manufacture of the dental instruments to ensure corrosion-resistance. In spite of this, the durability of these dental instruments
depends on their proper use and correct maintenance.
HANDLING OF THE BTR PEN
The stainless steel instrument is designed for multiple use, while the BTR PEN-compatible nitinol loop tip is intended for single use. Before each use, the instrument should be inspected for contamination, damage to the material structure (cracks, bends, splits, scratches – deeper scratches). Failure to adhere to the above stated instructions decreases instrument durability and reduces instrument lifetime.
HANDLING WITH BTR PEN NITINOL LOOP TIP
The BTR TIP is delivered non – sterile. Before use, the tip should be sterilized, using standard bags according to the sterilizaction in autoclave. Store in a dry and clean place for storage. Observe the instructions of the autoclave manufacturer. BTR TIP is for single use.
STERILIZE WITHOUT THE PROTECTIVE CUP!
Used tips disposed of responsibly or returned to the manufacturer.
HANDLING THE BTR PEN CALIBRATOR
The calibrator is designed to be reusable. It should be disinfected with isopropyl alcohol and inspected before each use. A blunted or bent calibrator needle can make it difficult to accurately calibrate the nitinol loop, which negatively affects the instrument performance.
CLEANING, RINSING AND WASHING OF BTR PEN:
The packaged instruments are non – sterile. Instruments that are brand new should be thoroughly cleaned with a non-scratching soft bristle brush and an instrument cleaning solution (detergent: ANIOSYME X3; solution prepared according to the manufacturer’s instructions). Ultrasonic cleaning is also recommended.
After cleaning, all components should be rinsed with purified water and dried, as moisture can adversely affect the effects of sterilisation. Instruments must be cleaned, rinsed and dried again and sterilised before each use on a subsequent patient.
STERILIZATION
All (reusable) dental instruments must be disinfected, then washed out and then dried before sterilisation. BTR PEN must always be disassembled before sterilisation. Instruments should be sterilized in accordance with national requirements.